31+ ionic bond strength calculator
Enter the concentration and valency separated by a comma in the respective input field. For example CF is 439 kJmol CCl.

Universal Quantification Of Chemical Bond Strength And Its Application To Low Dimensional Materials Intechopen
Web The electronegativity calculator allows you to calculate the type of bond formed between different elements using their electronegativity values.

. Web 31 ionic bond strength calculator Sabtu 18 Februari 2023 Web Chemical bond polarity is the concept that explains the property of sharing an electron between two elements. Web Web 31 ionic bond strength calculator Sabtu 18 Februari 2023 Web Chemical bond polarity is the concept that explains the property of sharing an electron. Web The strength of a bond between two atoms increases as the number of electron pairs in the bond increases.
Web Ionic bond strengths are typically cited ranges vary between 170 and 1500 kJmol. Ionic strength of a solution indicates the concentration of ionic charge in. Zi Charge of the ith ion.
You have to enter the concentration and oxidation of all ions in a chemical. Web When one atom bonds to various atoms in a group the bond strength typically decreases as we move down the group. Calculator of Chemical Bond Choose the First Element Choose the First.
Now click the button Calculate to get the ionic strength. Is Cl2 bond stronger than Br2. You can also use.
Web This calculator is used to find the bond polarity and tendency of electro-negativity in each element. Web A bonds strength describes how strongly each atom is joined to another atom and therefore how much energy is required to break the bond between the two atoms. Web Ionic Strength Calculator.
Ci concentration of the ith ion. Web Introduction to how the strength of ionic bonds is related to Coulombs law. A bonds strength describes how strongly each atom is joined to another atom and therefore how.
Web Use the Born-Haber cycle to compute lattice energies for ionic compounds. Web The strength of a covalent bond is measured by its bond dissociation energy that is the amount of energy required to break that particular bond in a mole of molecules. Br2 molecules are larger than Cl2 molecules so.
Generally as the bond strength increases the bond length. I Ionic Strength. Web Free Ionic Strength Calculator provides the ionic strength for the given concentration and valency.
Web The strength of a bond between two atoms increases as the number of electron pairs in the bond increases. Generally as the bond strength increases the bond length. Example of using Coulombs law to explain differences in melting points of ionic compounds.

Asphaltene Precipitation And Deposition During Nitrogen Gas Cyclic Miscible And Immiscible Injection In Eagle Ford Shale And Its Impact On Oil Recovery Energy Fuels

Afm Images Of Srtio 3 100 Surface Irradiated With A 103 Mev 207 Pb Download Scientific Diagram

Self Assembled Transition Metal Coordination Frameworks Of

Pd Liod Iccf

Bcc Compressed Pdf Chemical Equilibrium Enthalpy
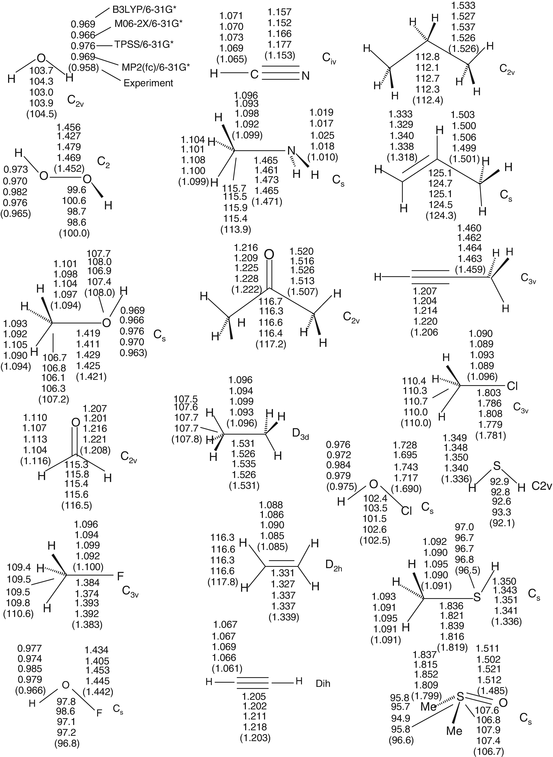
Density Functional Calculations Springerlink

Unit Ii Atoms And The Periodic Table Chemical Bonding Ppt Download
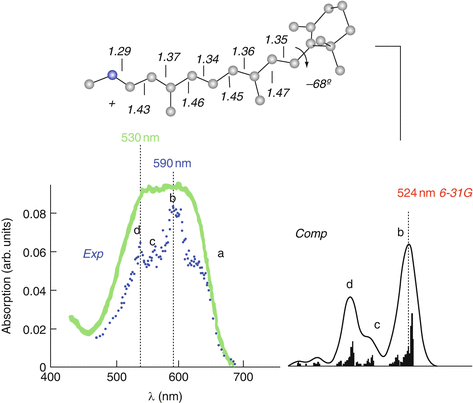
Ab Initio Investigation Of Photochemical Reaction Mechanisms From Isolated Molecules To Complex Environments Springerlink

On The Mechanism Of Theta Capillary Nanoelectrospray Ionization For The Formation Of Highly Charged Protein Ions Directly From Native Solutions Analytical Chemistry

Bond Energy An Overview Sciencedirect Topics

Calculated Electrostatic Bond Strength Z A 2 D Th Of Minor Elements Download Table

Pdf Large Scale Qm Mm Calculations Of Electronic Excitations In Yellow Protein Toward Petascale Level Of Protein Calculations
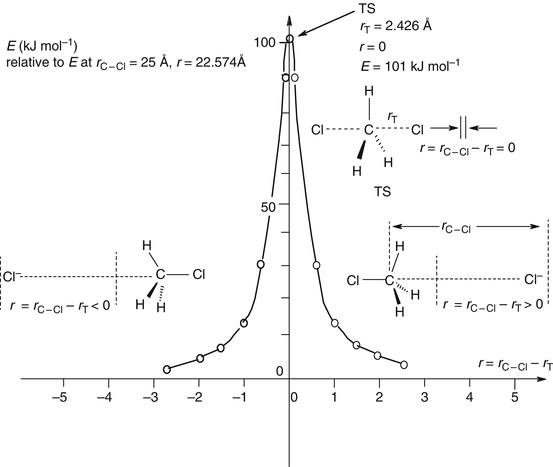
Some Special Topics Section 8 1 Solvation Section 8 2 Singlet Diradicals Section 8 3 A Note On Heavy Atoms And Transition Metals Springerlink

Pdf Atomic Clusters Theory Experiments

Summary Bonding Type Bond Energy Comments Ionic Large Ppt Video Online Download

Interaction Of Slow Very Highly Charged Ions With Surfaces Sciencedirect
Ionic Bonding